Mitochondrial distribution and interactions with microtubules at cell division dictates growth potential to daughter cells in fission yeast.
In symmetric cell division, the contents of mother cells are divided equitably to ensure that daughter cells have the same potential for growth toward the next cell cycle.
Scientists from UNSW Sydney’s School of Biomedical Sciences, with their international colleagues, have now uncovered the mechanism – involving crucial interactions between two cellular components – that facilitates symmetric cell division.
Their experiments revealed that binding of mitochondria to microtubules was crucial in this process.
Microtubules are intracellular networks that maintain the structure and shape of a cell, while also serving as a highway for transport of proteins and other cargo.
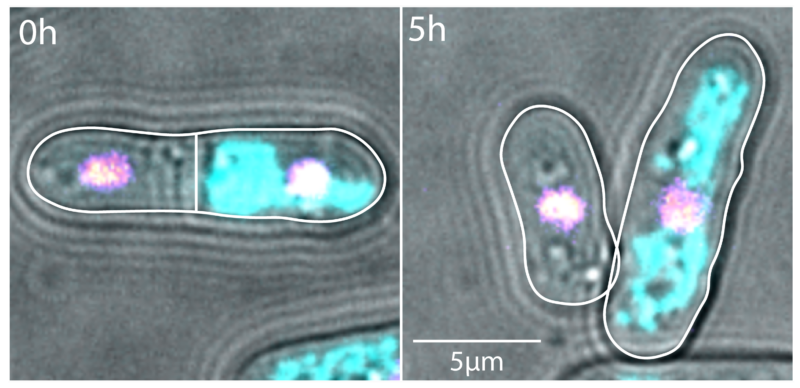
The researchers report, in the Journal of Cell Science, how the interplay between microtubules and mitochondria needs to be within a ‘Goldilocks zone’ to ensure symmetrical cell division.
The consequence of asymmetric cell division is not only one bigger and one smaller daughter cell, but also uneven distribution of mitochondrial DNA from the mother cell.
“We show very clear evidence of microtubule dynamics changing when you have mitochondria bound to them. And microtubule dynamics are inherently responsible for ensuring mitochondrial lengths are evenly distributed within the cell,” said Dr Vaishnavi Ananthanarayanan, EMBL Australia Group Leader and the study’s senior author.
Mitochondria are the cell’s energy powerhouses, so the researchers were not surprised to see that the cells growing the fastest were the bigger ones that received more copies of mitochondria than their smaller, slower growing sister cells. Mitochondria are inherited from mother cells and the numbers do not change during cell growth.
With live cell imaging, the research team unravel the process that determines the destiny of daughter cells, using fission yeast as a model system.
Mechanisms regulating symmetrical cell division
In fission yeast, the positioning of the nucleus just before the cell enters division determines where it will be divided. Dr Ananthanarayanan explains how microtubules are responsible for centring the nucleus
before the cell enters mitosis.
“The nuclear position is determined by microtubules which emanates close to the nuclear envelope. Microtubules on either side of the cell grow out and push against the cell wall, pushing the nucleus in the opposite direction. The force balance is maintained since there are the same number of microtubules on each side, and hence the nucleus remains roughly at the centre of the cell,” she said.
“If you now don’t have symmetric cell division, you end up not having partitioning of mitochondria that is equitable between the daughter cells.”
Fission yeast typically divide symmetrically. By introducing mutations to alter mitochondrial attachment to microtubules – causing destabilisation – the researchers gained a molecular understanding of cell division and got a glimpse of what can happen when these finely balanced processes go awry in diseased cells.
Dr Ananthanarayanan explains that this study originated from observations made during a previous related project by a Ph.D. candidate in her group, Ms Leeba Ann Chacko.
Lead author of the current study, Ms Chacko had noticed that a significant proportion of cells with mutant forms of mitochondria were dividing asymmetrically.
“I was also very excited when I first observed that the nucleus is more dynamic in cells with fewer mitochondria,” said Ms Chacko in her first-person interview with Journal of Cell Science.
While the research was conducted in yeast cells, the researchers believe their findings are likely to be recapitulated in mammalian cell, which have similar microtubule and mitochondria organisation. And they have already begun to verify these new findings in mammalian cells.
Read the full Journal of Cell Science article here.